Have you ever noticed how milk turns into curd or how iron slowly gets rusted? These are not just random changes—they are chemical reactions happening around us every day.
In this chapter, we’ll understand how these reactions occur and how scientists represent them using equations. Let’s begin
Table of Contents
What is Chemical Reaction? Easy Explanation
A chemical reaction is a process where the original substances (known as reactants) are transformed into entirely new substances (known as products) through the rearrangement of atoms.
In easy language we said that ‘A process in which one or more substances change into new substances with different properties’. Example:
Magnesium + Oxygen → Magnesium oxide
Let’s understand this with a real-life example: Making curd from milk, Rusting of iron, Food digestion, burning candle etc. These are all examples of chemical reactions.
How to identify a chemical reaction?
You can observe that a chemical reaction has taken place by looking for one or more of the following signs:
| Observation | Meaning |
|---|---|
| Change in colour | New substance formed |
| Change in temperature | Heat released/absorbed |
| Gas formation | Bubbles/smoke appearing |
| Change in state | Solid/liquid/gas change |
Magnesium Ribbon Example: When you burn a magnesium ribbon in air, it reacts with oxygen to form a white powder called Magnesium Oxide (MgO).

What is a Chemical Equation?
Chemical Equation is a short representation of a chemical reaction using symbols or words. Example: Mg + O₂ → MgO
Substances that react (Left side) it’s called Reactants, Substances formed (Right side) it’s called Products and Arrow (→) sign shows direction of the equation.
But right now, this is a Skeletal Equation. A skeletal equation is a chemical equation written using formulas, but it is not balanced. That means:
Number of atoms on left side ≠ number of atoms on right side
| Element | Left Side | Right Side |
|---|---|---|
| Mg | 1 | 1 |
| O | 2 | 1 |
Upon counting the atoms here, we found that the oxygen count is unequal; therefore, the equation is unbalanced. So, this is called a skeletal (unbalanced) equation.
Why is it Important?
A skeletal equation is just the first step. After writing it, we:
- Balance the equation
- Make atoms equal on both sides
Balanced Form: 2Mg + O₂ → 2MgO
Now: Mg = 2 on both sides, O = 2 on both sides (Equation is now balanced)
To represent these reactions on paper, we use chemical equations. There are some main components shown in the image below
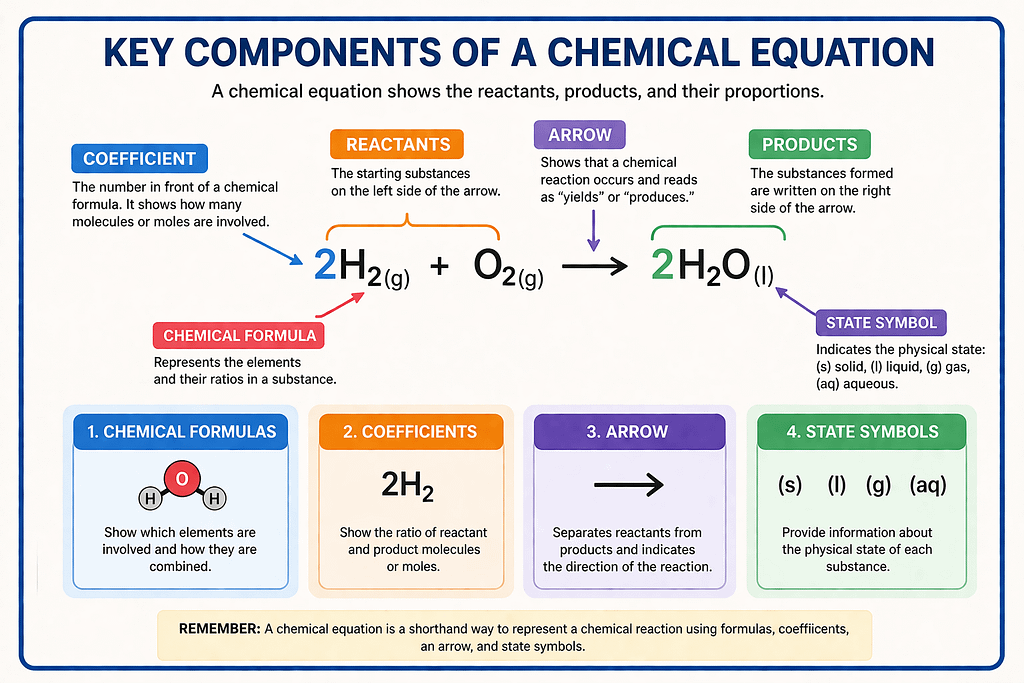
Balanced Chemical Equations
A chemical equation in which the number of atoms of each element is equal on both sides called balanced Chemical Equation.
According to the Law of Conservation of Mass (mass cannot be created or destroyed), we must ensure the number of atoms is equal on both sides. Example:
Zn + H₂SO₄ → ZnSO₄ + H₂ (Balanced)
How to balance a chemical Equation?
Balancing a chemical equation means making sure the number of atoms of each element is equal on both sides.
The hit & trial method is the simplest way to do this—just adjust atoms step by step until everything matches.
Step 1: Write the Skeletal Equation
Start with the unbalanced equation:
Fe + H₂O → Fe₃O₄ + H₂
This is your starting point (atoms are not equal yet).
Step 2: Count Atoms on Both Sides
| Element | Left Side | Right Side |
|---|---|---|
| Fe | 1 | 3 |
| H | 2 | 2 |
| O | 1 | 4 |
Step 3: Balance One Element at a Time
(a) Balance Iron (Fe):
Right side has 3 Fe → put 3 before Fe
3Fe + H₂O → Fe₃O₄ + H₂
(b) Balance Oxygen (O):
Right side has 4 O → we need 4 O on left
Each H₂O has 1 O → put 4 before H₂O
3Fe + 4H₂O → Fe₃O₄ + H₂
(c) Balance Hydrogen (H):
Left side: 4H₂O = 8 H
Right side: H₂ = 2 H
To make 8 H on right, put 4 before H₂
3Fe + 4H₂O → Fe₃O₄ + 4H₂
Step 4: Check Final Equation
| Element | Left Side | Right Side |
|---|---|---|
| Fe | 3 | 3 |
| O | 4 | 4 |
| H | 8 | 8 |
All atoms are equal. So, the equation is balanced
Final Balanced Equation:
3Fe + 4H₂O → Fe₃O₄ + 4H₂
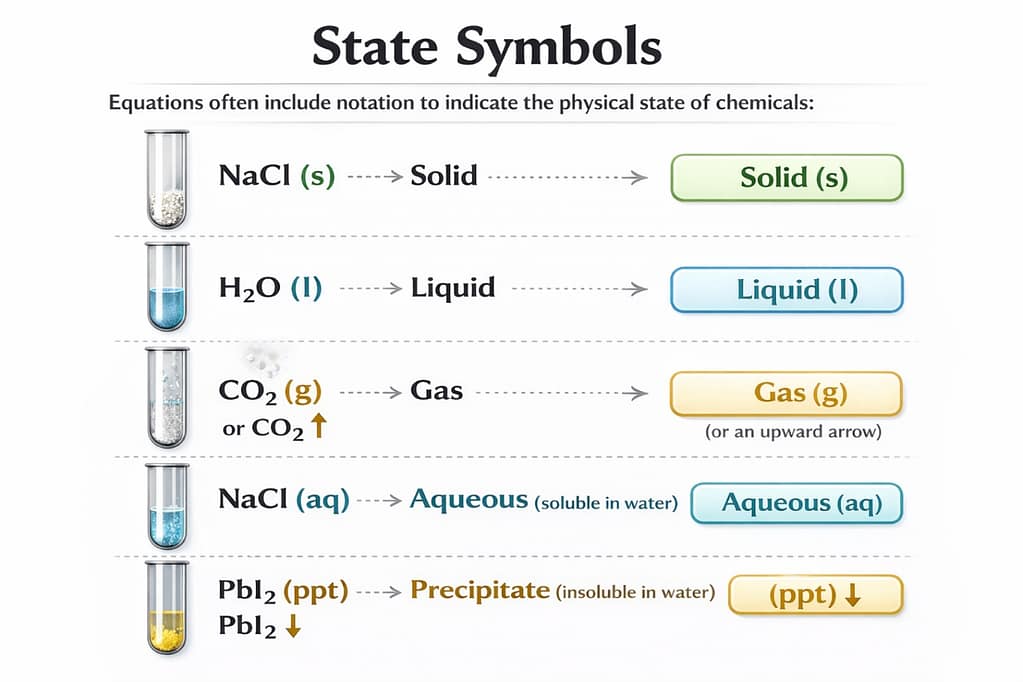
Why are state symbols important?
They make the chemical equation more clear and informative by showing the physical state of reactants and products. Understand State symbols with image 👇
Types of Chemical Reactions
Understanding how substances interact is the core of this chapter, Let’s find out how many types of chemical reactions there are:
- Combination reaction
- Decomposition reaction
- Displacement reaction
- Double displacement reaction
- Exothermic and Endothermic
- Oxidation and Reduction (Redox)
Combination Reaction: A combination reaction (also known as a synthesis reaction) occurs when two or more substances (reactants) combine to form a single new substance (product). This is represented by the general equation: A + B -> AB.
CaO + H₂O → Ca(OH)₂
if you want to Master this chapter, click the download button provided below 👇 understand this entire chapter in simple language.
Download Chemical Reactions and Equations Notes PDF

important words..
Now, you must have thoroughly understood this entire chapter. If you can explain this chapter all concepts to yourself, then you are ready for the Test series (Quiz). And yes! Don’t forget to read the important FAQs. If you have any questions or suggestions leave your comment.
Chemical Reactions and Equations MCQ Quiz
Why should a magnesium ribbon be cleaned before burning in air ?
Magnesium is a very reactive metal. When stored, it reacts with oxygen to form a protective layer of basic magnesium carbonate on its surface. This layer prevents further reaction with oxygen. Cleaning it with sandpaper removes this layer so the ribbon can burn properly.
What is a “skeletal” chemical equation, and why must we balance it ?
A skeletal equation is an unbalanced chemical equation that represents a reaction using formulas but does not show equal atoms on both sides. It must be balanced to satisfy the Law of Conservation of Mass, which states that mass (and therefore atoms) cannot be created or destroyed in a chemical reaction.
Why is respiration considered an exothermic reaction ?
During respiration, glucose (C_6H_{12}O_6) combines with oxygen in our body cells to produce carbon dioxide and water. This process releases a significant amount of energy, and reactions that release energy are called exothermic reactions.
Why are oil and fat-containing food items flushed with nitrogen ?
Nitrogen is an inert (unreactive) gas. When food containing fats and oils is exposed to oxygen, it undergoes oxidation, leading to rancidity (bad smell and taste). Flushing the packet with nitrogen prevents oxidation and keeps the food fresh.
